|
5/30/2023 0 Comments Ion bonding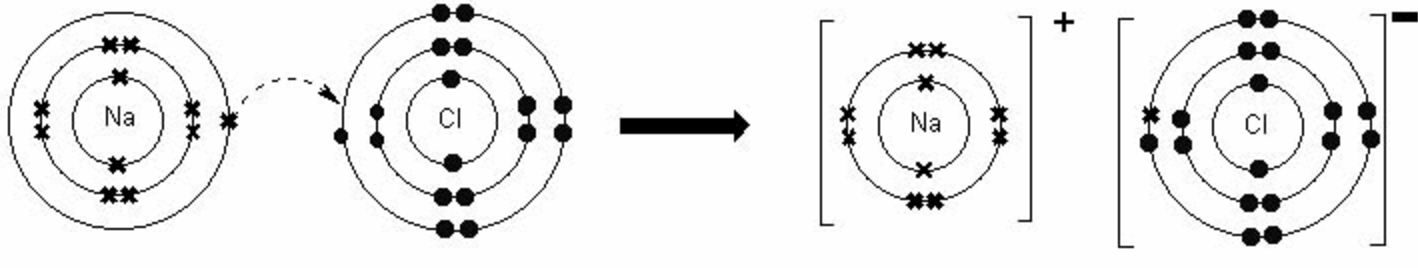
Toward the upper right hand corner of the periodic table because of aĬombination of nuclear charge and shielding factors. How evenly a pair of electrons in a bond is shared. Looking at the electronegativity values of different atoms helps us to decide There is more negative charge toward one end of theīond, and that leaves more positive charge at the other end. 
In a polar covalent bond, a pair of electrons is shared between twoĪtoms in order to fulfill their octets, but the electrons lie closer to one end There is not a simple answer to this question. How can you tell if a compound is ionic or covalent? Ions are used to maintain cell potentials andĪre important in cell signaling and muscle contraction. For example, there are many different ionicĬompounds (salts) in cells. One of the roles of the water is toĭissolve different materials. Which bonds are ionic and which are covalent?Ĭells contain lots of water.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
